The support provided is really helpful and I
never had a prescription medication company
follow up this way. It is pretty amazing!
– Real patient with Parkinson's disease
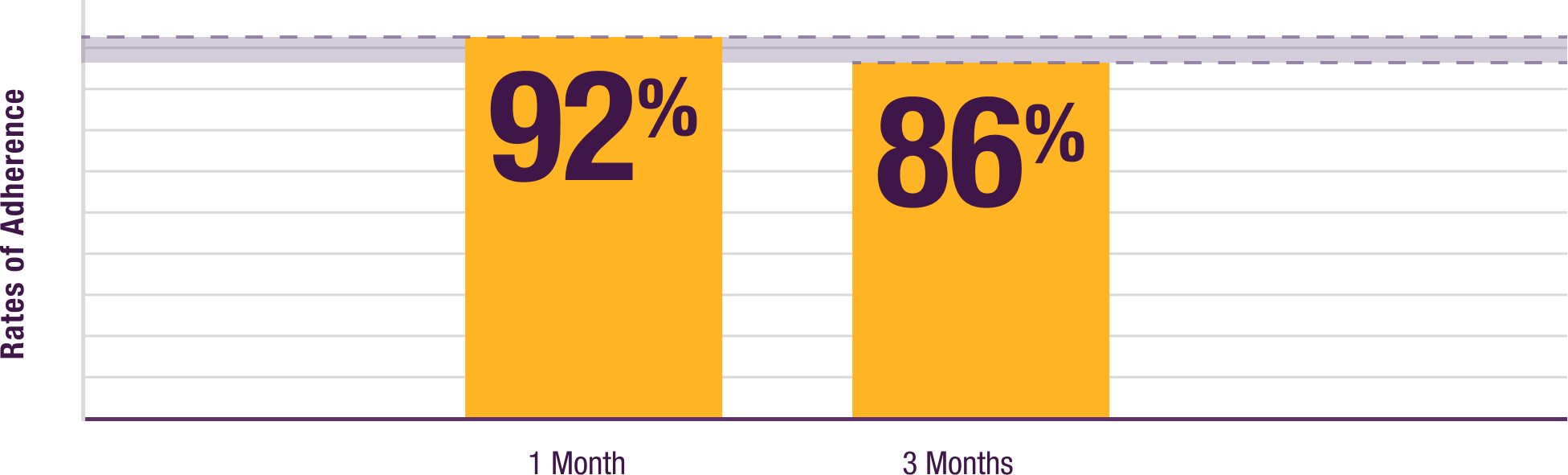
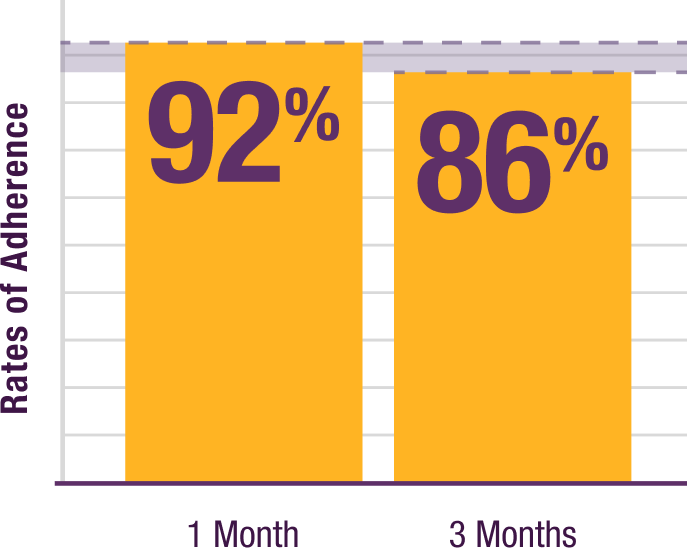
Patients enrolled in a Nurse Education program maintained
a consistent adherence rate over time after training22