Administered via the pulmonary route, INBRIJA complements oral CD/LD treatment
-
Delivers consistent absorption14
-
Bypasses problems associated with the GI tract14
-
Allows for on-demand use. See dosing information
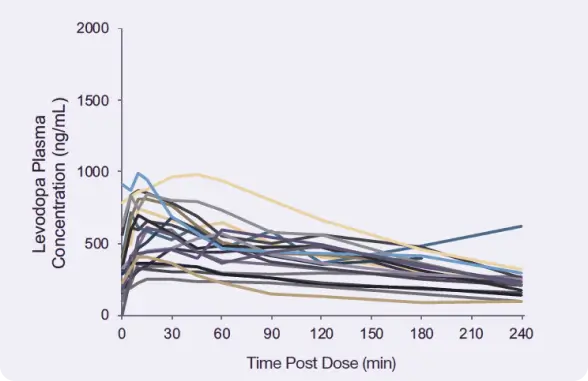
Absorption of levodopa after a high-fat, high-protein meal14,*
INBRIJA 84 mg† (n=20)
Oral CD/LD 25/100 mg (n=17)
.webp)
Results from a randomized, open-label, 2-way crossover study evaluating the pharmacokinetics (PK) of a single inhaled dose of INBRIJA 84 mg, administered with oral carbidopa (CD) 25 mg, and a single ingested dose of CD/LD 25/100 mg in fed state in patients with PD who were regularly taking oral CD/LD.*
Mean LD Plasma Concentrations After a Dose of INBRIJA 84 mg or Oral CD/LD 25/100 mg Following a
High-Fat, High-Protein Meal*

Increase in mean plasma levels within 5 minutes

Median Tmax compared with
120 minutes for oral CD/LD‡

*4 to 5 hours after morning CD/LD, patients ate a meal and then received study drug.
†INBRIJA 84 mg coadministered with 25 mg oral carbidopa for true PK comparison to levodopa.
‡Median Tmax represented to account more accurately for outliers.
SD, standard deviation; Tmax, time to peak drug concentration.


 Please see Full Prescibing
Please see Full Prescibing